Ankit Chawla, Amite Pankaj Aggarwal
Volume 1 | Issue 2 | Aug – Nov 2016 | Page 3-7
Author: Ankit Chawla[1], Amite Pankaj Aggarwal[1]
[1] Unit of Joint Replacement, Arthroscopy and Orthopaedics, Fortis Hospital, Shalimar Bagh, New Delhi, India.
Address of Correspondence
Dr. Amite Pankaj Aggarwal
Fortis Hospital Shalimar Bagh
New Delhi, India.
Email: amitepankaj@gmail.com.
Abstract
Meniscal injuries are recognized as a cause of significant musculoskeletal morbidity. The menisci are vital for the normal function and long-term health of the knee joint. And loss of a meniscus increases the risk of subsequent development of degenerative changes in the knee. A review of anatomy and ultrastructure of the meniscus, and its relationship to normal function in terms of load transmission, shock absorption, joint stability, lubrication and nutrition is a necessary prerequisite to understanding pathologies associated with the knee.
Keywords: Meniscus, Medial meniscus, lateral meniscus, Anatomy, Function.
Introduction
The word meniscus comes from the Greek word me-niskos, meaning “crescent,” diminutive of me-ne-, meaning “moon.” The menisci are semilunar discs of fibrocartilaginous tissue which are vital for the normal biomechanics and long-term health of the knee joint [1]. The characteristic shape of the lateral and medial menisci is attained between the 8th and 10th week of gestation. They arise from a condensation of the intermediate layer of mesenchymal tissue to form attachments to the surrounding joint capsule[2,3].
Gross Anatomy
These crescent-shaped wedges of fibrocartilage are located on the medial and lateral aspects of the knee joint (Fig. 1A,1B). The peripheral, vascular border of each meniscus is thick, convex, and attached to the joint capsule. The innermost border tapers to a thin free edge. The superior surfaces of menisci are concave, enabling effective articulation with their respective convex femoral condyles. The inferior surfaces are flat to accommodate the tibial plateau [4,5].
Medial Meniscus
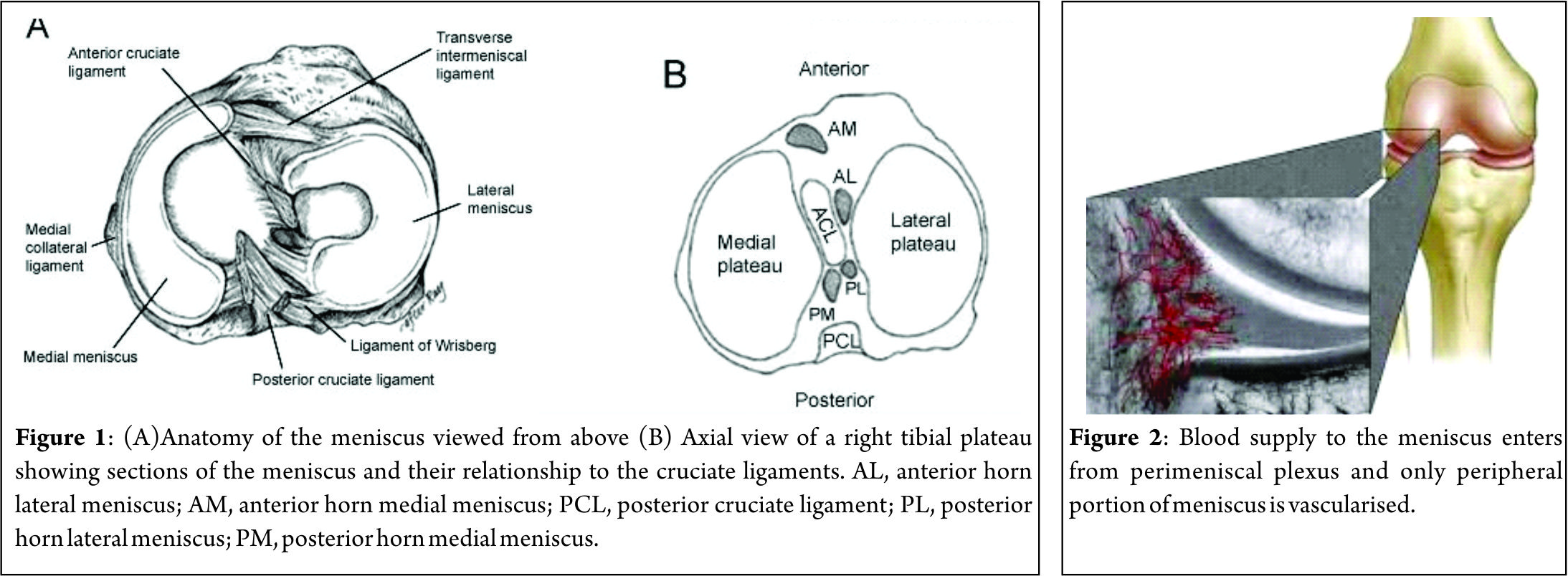
The medial meniscus is a C-shaped structure larger in radius than the lateral meniscus, with the posterior horn being wider than the anterior. The anterior horn is attached firmly to the tibia anterior to the intercondylar eminence and to the anterior cruciate ligament. The posterior horn is anchored immediately in front of the attachments of the posterior cruciate ligament posterior to the intercondylar eminence. Its entire peripheral border is firmly attached to the medial capsule and through the coronary ligament to the upper border of the tibia. At its midpoint, the medial meniscus is more firmly attached to the femur through a condensation in the joint capsule known as the deep medial collateral ligament [5]. The transverse, or “intermeniscal,” ligament is a fibrous band of tissue that connects the anterior horn of the medial meniscus to the anterior horn of the lateral meniscus [5,6].
Lateral Meniscus
The lateral meniscus is more circular in form, covering up to two thirds of the articular surface of the underlying tibial plateau [7]. The anterior horn is attached to the tibia medially in front of the intercondylar eminence, whereas the posterior horn inserts into the posterior aspect of the intercondylar eminence and in front of the posterior attachment of the medial meniscus. The lateral meniscus is loosely attached to the capsular ligament; however, these fibers do not attach to the lateral collateral ligament. The posterior horn of the lateral meniscus attaches to the inner aspect of the medial femoral condyle via the anterior and posterior meniscofemoral ligaments of Humphrey and Wrisberg, respectively, which originate near the origin of the PCL (Fig. 1A) [8]. Their estimated prevalence is 74 % for Humphrey ligament, 69 % for Wrisberg ligament, and both ligaments found together in around 50 % of knees [9]. The lateral meniscus is smaller in diameter, thicker in periphery, wider in body, and more mobile than the medial meniscus.
Extracellular matrix and cellularity
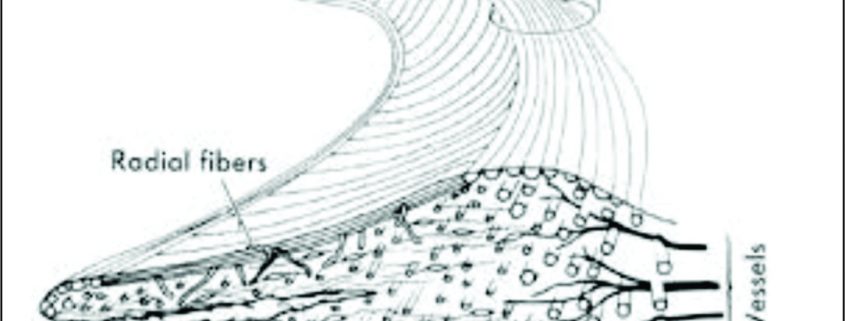
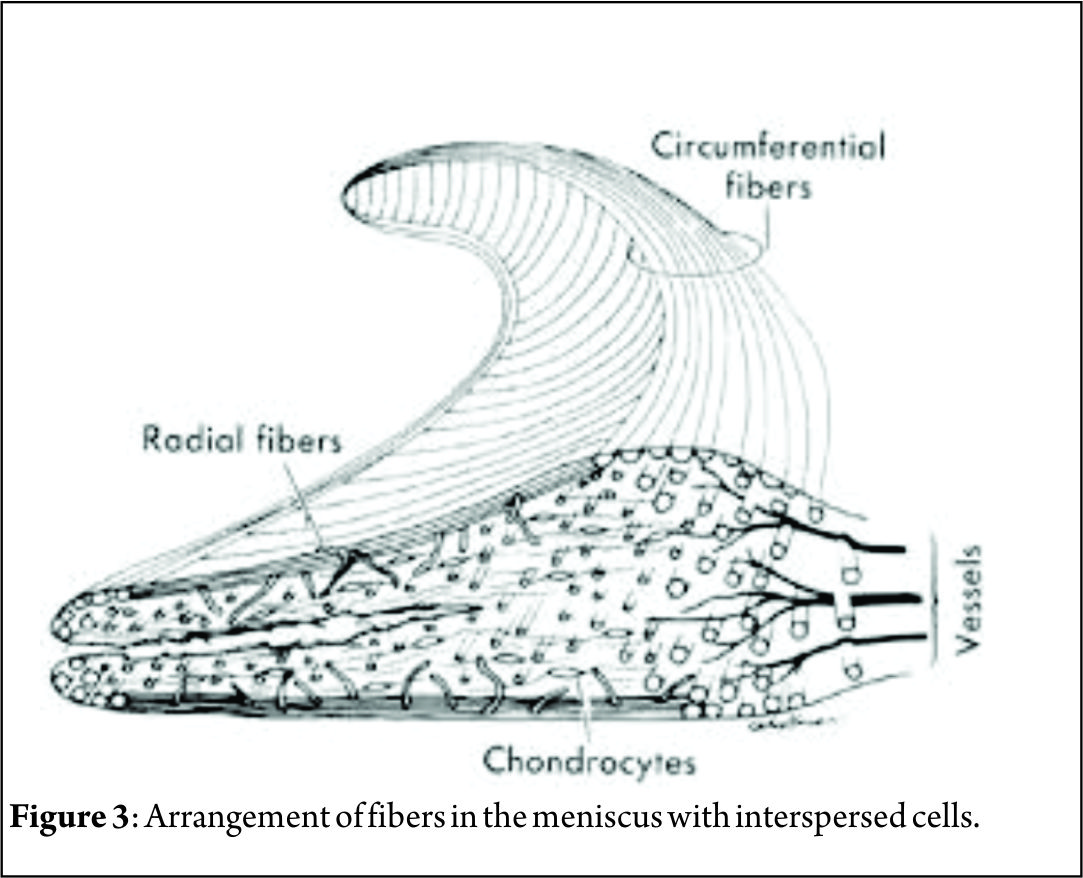
Considering composition by wet weight, the meniscus has high water content (72 %). The remaining 28 % consists of an organic component, mostly ECM and cells.10 Collagens comprise the majority (75 %) of the organic matter, followed by GAGs (17 %), DNA (2 %), adhesion glycoproteins (<1 %), and elastin (<1 %) [10,11]. These proportions vary according to age, injury, or pathological conditions [12]. Collagen is the main fibrillar component of the meniscus. Different collagen types exist in various quantities in each region of meniscus. In the red–red zone, type I collagen is predominant (80 % composition in dry weight). In the white–white zone, 60 % is type II collagen and 40 % is type I collagen [13]. The major orientation of collagen fibers in the meniscus is circumferential; radial fibers and perforating fibers also are present.(Fig. 3) [13]. Proteoglycans are heavily glycosylated molecules that constitute a major component of the meniscus ECM [14]. These molecules are comprised of a core protein which is decorated with glycosaminoglycans (GAGs). The main types of GAGs found in normal human meniscal tissue are chondroitin 6 sulfate (60%), dermatan sulfate(20-30%), chondroitin 4 sulfate (10-20%), and keratin sulfate(15%) [15]. Their main function is to enable the meniscus to absorb water, whose confinement supports the tissue under compression [10]. Adhesion glycoproteins are also important components of the meniscus matrix, as they serve as a link between ECM components and cells [16]. The main adhesion glycoproteins present in the human meniscus are fibronectin, thrombospondin, and collagen VI [16,17]. Outer zone cells have an oval, fusiform shape and are similar in appearance and behaviour to fibroblasts, described as fibroblast-like cells [18]. The matrix surrounding the cells is mainly comprised of type I collagen, with small percentages of glycoproteins and collagen types III and V present. In contrast, cells in the inner portion have rounded appearance and are embedded in an ECM comprising largely type II collagen intermingled with a smaller but significant amount of type I collagen and higher concentration of GAGs [18]. This relative abundance of collagen type II and aggrecan in the inner region is more reminiscent of hyaline articular cartilage. Therefore, cells in this region are classified as fibrochondrocytes or chondrocyte like cells. In summary, cell phenotype and ECM composition render the outer portion of the meniscus akin to fibrocartilage, while the inner portion possesses similar, but not identical, traits to articular cartilage [19,20].
Vascularity and Innervation
The vascular supply to the medial and lateral menisci originates predominantly from the lateral and medial geniculate vessels (both inferior and superior). Branches from these vessels give rise to a perimeniscal capillary plexus within the synovial and capsular tissue. (Fig. 2) Radial branches from the plexus enter the meniscus at intervals, with a richer supply to the anterior and posterior horns. Vessels supplying the body are limited to the meniscus periphery with a variable penetration of 10–30 % for medial meniscus and 10–25 % for lateral one. This has important implication for meniscal healing [21]. The remaining portion of each meniscus (65% to 75%) receives nourishment from synovial fluid via diffusion or mechanical pumping (ie, joint motion) [22, 23]. The knee joint is innervated by the posterior articular branch of the posterior tibial nerve and the terminal branches of the obturator and femoral nerves. The lateral portion of the capsule is innervated by the recurrent peroneal branch of the common peroneal nerve. These nerve fibers penetrate the capsule and follow the vascular supply to the peripheral portion of the menisci and the anterior and posterior horns, where most of the nerve fibers are concentrated. The inner menisci core has no nerve fibers [21].
Biomechanical Function
The biomechanical function of the meniscus is a reflection of the gross and ultrastructural anatomy and of its relationship to the surrounding intra-articular and extra-articular structures. The meniscus withstands many different forces such as shear, tension, and compression. It also plays a crucial role in load-bearing, load transmission, shock absorption, stability, propioception as well as lubrication and nutrition of articular cartilage [24-27]. They also serve to decrease contact stresses and increase contact area and congruity of the knee [28,29].
Meniscal Biomechanics
The biomechanical properties of the knee meniscus are appropriately tuned to withstand the forces exerted on the tissue. Many studies have helped to quantify the properties of the tissue both in humans and in animal models. According to these studies, the meniscus resists axial compression with an aggregate modulus of 100-150 kPa [30]. The tensile modulus of the tissue varies between the circumferential and radial directions; it is approximately 100-300 MPa circumferentially and 10 fold lower than this radially [31]. Finally, the shear modulus of the meniscus is approximately 120 kPa [31]. The contact forces on the meniscus within the human knee joint have been mapped. It has been calculated that the intact menisci occupy approximately 60% of the contact area between the articular cartilage of the femoral condyles and the tibial plateau, while they transmit >50% of the total axial load applied in the joint [32,33]. However, these percentages are highly dependent on degree of knee flexion and tissue health. For every 30o of knee flexion, the contact surface between the two knee bones decreases by 4% [34]. When the knee is in 90o of flexion the applied axial load in the joint is 85% greater than when it is in 0o of flexion [33]. In full knee flexion, the lateral meniscus transmits 100% of the load in the lateral knee compartment, whereas the medial meniscus takes on approximately 50% of the medial load [29]. Studies confirm that there is a significant difference in segmental motion during flexion between the medial and lateral menisci. The anterior and posterior horn lateral meniscus ratio is smaller and indicates that the meniscus moves more as a single unit [35]. Alternatively, the medial meniscus (as a whole) moves less than the lateral meniscus, displaying a greater anterior to posterior horn differential excursion. Thompson et al found that the area of least meniscal motion is the posterior medial corner, where the meniscus is constrained by its attachment to the tibial plateau by the meniscotibial portion of the posterior oblique ligament, which has been reported to be more prone to injury [35,36]. A reduction in the motion of the posterior horn of the medial meniscus is a potential mechanism for meniscal tears, with a resultant “trapping” of the fibrocartilage between the femoral condyle and the tibial plateau during full flexion. The greater differential between anterior and posterior horn excursion may place the medial meniscus at a greater risk of injury [35]. The differential of anterior horn to posterior horn motion allows the menisci to assume a decreasing radius with flexion, which correlates to the decreased radius of curvature of the posterior femoral condyles [35]. This change of radius allows the meniscus to maintain contact with the articulating surface of both the femur and the tibia throughout flexion.
Load Transmission
Fairbank described the increased incidence and predictable degenerative changes of the articular surfaces in completely meniscectomized knees [37]. Weightbearing produces axial forces across the knee, which compress the menisci, resulting in “hoop” (circumferential) stresses [38]. Hoop stresses are generated as axial forces and converted to tensile stresses along the circumferential collagen fibers of the meniscus. Firm attachments by the anterior and posterior insertional ligaments prevent the meniscus from extruding peripherally during load bearing [39]. Medial meniscectomy decreases contact area by 50% to 70% and increases contact stress by 100%. Lateral meniscectomy decreases contact area by 40% to 50% but dramatically increases contact stress by 200% to 300% because of the relative convex surface of the lateral tibial plateau [40,41]. This significantly increases the load per unit area and may contribute to accelerated articular cartilage damage and degeneration [42].
Shock absorption
The menisci play a vital role in attenuating the intermittent shock waves generated by impulse loading of the knee with normal gait [43,44]. Voloshin and Wosk showed that the normal knee has a shock-absorbing capacity about 20% higher than knees that have undergone meniscectomy [38]. As the inability of a joint system to absorb shock has been implicated in the development of osteoarthritis, the meniscus would appear to play an important role in maintaining the health of the knee joint [45]
Joint stability
The geometric structure of the menisci provides an important role in maintaining joint congruity and stability. The superior surface of each meniscus is concave, enabling effective articulation between the convex femoral condyles and flat tibial plateau. When the meniscus is intact, axial loading of the knee has a multidirectional stabilizing function, limiting excess motion in all directions [46]. The studies for effects of meniscectomy on joint laxity for anteroposterior and varus-valgus motions and rotation have indicated indicated that the effect on joint laxity depends on whether the ligaments of the knee are intact and whether the joint is bearing weight. In the presence of intact ligamentous structures, excision of the menisci produces small increases in joint laxity. In an anterior cruciate ligament–deficient knee, medial meniscectomy has been shown to increase tibial translation by 58% at 90o, whereas primary anterior and posterior translations were not affected by lateral meniscectomy [47]. Shoemaker and Markolf demonstrated that the posterior horn of the medial meniscus is the most important structure resisting an anterior tibial force in the ACL-deficient knee. [48] Recently, Musahl et al reported that the lateral meniscus plays a role in anterior tibial translation during the pivot-shift maneuver [49].
Joint Nutrition and Lubrication
The menisci may also play a role in the nutrition and lubrication of the knee joint. The mechanics of this lubrication remains unknown; the menisci may compress synovial fluid into the articular cartilage, which reduces frictional forces during weightbearing [50]. There is a system of microcanals within the meniscus located close to the blood vessels, which communicates with the synovial cavity; these may provide fluid transport for nutrition and joint lubrication [51,52].
Conclusions
Mechanoreceptors have been identified in the anterior and posterior horns of the menisci, middle and outer third of the meniscus. The identification of these neural elements indicates that the menisci are capable of detecting proprioceptive information (joint motion and position) in the knee joint, thus playing an important afferent role in the sensory feedback mechanism of the knee [53,54].
References
1. McDermott ID, Masouros SD, Amis AA. Biomechanics of the menisci of the knee. Curr Orthopaed.2008 ;22:193–201
2. Gardner E, O’Rahilly R. The early development of the knee joint in staged human embryos. J Anat.1968;102:289-299.
3. Gray DJ, Gardner E. Pre-natal development of the human knee and superior tibial fibula joints. Am J Anat.1950;86:235-288.
4. Bullough PG, Vosburgh F, Arnoczky SP, et al. The menisci of the knee. In: Insall JN, ed. Surgery of the Knee. New York, NY: Churchill Livingstone; 1984:135-149.
5. Warren RF, Arnoczky SP, Wickiewiez TL. Anatomy of the knee. In: Nicholas JA, Hershman EB, eds. The Lower Extremity and Spine in Sports Medicine. St. Louis: Mosby; 1986:657-694.
6. Johnson DL, Swenson TD, Harner CD. Arthroscopic meniscal transplantation: anatomic and technical considerations. Presented at: Nineteenth Annual Meeting of the American Orthopaedic Society for Sports Medicine; July 12-14, 1993; Sun Valley, ID.
7. Arnoczky SP, Adams ME, DeHaven KE, Eyre DR, Mow VC. The meniscus. In: Woo SL-Y, Buckwalter J, eds. Injury and Repair of Musculoskeletal Soft Tissues. Park Ridge, IL: American Academy of Orthopaedic Surgeons; 1987:487-537.
8. Johnson DL, Swenson TM, Livesay GA, Aizawa H, Fu FH, Harner CD. Insertion-site anatomy of the human menisci: gross, arthroscopic, and topographical anatomy as a basis for meniscal transplantation. Arthroscopy.1995;11:386-394.
9. Heller L, Langman J. The meniscofemoral ligaments of the human knee. J Bone Joing Surg Br.1964;46 :307-313.
10. Makris EA, Hadidi P, Athanasiou KA. The knee meniscus: structure-function, pathophysiology, current repair techniques, and prospects for regeneration. Biomaterials. 2011 Oct;32(30):7411-31.
11. Herwig J, Egner E, Buddecke E. Chemical changes of human knee joint menisci in various stages of degeneration. Ann Rheum Dis. 1984 Aug;43(4):635-40.
12. Sweigart MA, Athanasiou KA. Toward tissue engineering of the knee meniscus. Tissue Eng. 2001 Apr;7(2):111-29.
13. Cheung HS. Distribution of type I, II, III and V in the pepsin solubilized collagens in bovine menisci. Connect Tissue Res. 1987;16(4):343-56.
14. Beaupre A, Choukroun R, Guidouin R, Carneau R, Gerardin H. Knee menisci: correlation between microstructure and biomechanics. Clin Orthop Relat Res.1986 ;208 :72-75.
15. Assimakopoulos AP, Katonis PG, Agapitos MV, Exarchou EI. The innervations of the human meniscus. Clin Orthop Relat Res.1992;275:232-236.
16. Miller RR, McDevitt CA. Thrombospondin in ligament, meniscus and intervertebral disc. Biochim Biophys Acta. 1991 Nov 14;1115(1):85-8
17. McDevitt CA, Webber RJ. The ultrastructure and biochemistry of meniscal cartilage. Clin Orthop Relat Res. 1992; 252:8–18.
18. Verdonk PC, Forsyth RG, Wang J et al (2005) Characterisation of human knee meniscus cell phenotype. Osteoarthritis Cartilage 13:548–560.
19. Melrose J, Smith S, Cake M, Read R, Whitelock J. Comparative spatial and temporal localisation of perlecan, aggrecan and type I, II and IV collagen in the ovine meniscus: an ageing study. Histochem Cell Biol. 2005; 124:225–35.
20. Hellio Le Graverand MP, Ou Y, Schield-Yee T, Barclay L, Hart D, Natsume T, et al. The cells of the rabbit meniscus: their arrangement, interrelationship, morphological variations and cytoarchitecture. J Anat. 2001; 198:525–35.
21. Brian D, Mackenzie WG, Shim SS, Leung G (1985) The vascular and nerve supply of the human meniscus. Arthroscopy 1:58–62.
22. Meyers E, Zhu W, Mow V. Viscoelastic properties of articular cartilage and meniscus. In: Nimni M, ed. Collagen: Chemistry, Biology and Biotechnology. Boca Raton, FL: CRC; 1988.
23. Mow V, Fithian D, Kelly M. Fundamentals of articular cartilage and meniscus biomechanics. In: Ewing JW, ed. Articular Cartilage and Knee Joint Function: Basic Science and Arthroscopy New York, NY: Raven Press; 1989:1-18.
24. Cameron HU, Macnab I. The structure of the meniscus of the human knee joint. Clin Orthop Relat Res. 1972; 89:215–9.
25. Newman AP, Anderson DR, Daniels AU, Dales MC. Mechanics of the healed meniscus in a canine model. Am J Sports Med. 1989; 17:164–75.
26. Zhu W, Chern KY, Mow VC. Anisotropic viscoelastic shear properties of bovine meniscus. Clin Orthop Relat Res. 1994; 306:34–45.
27. Tissakht M, Ahmed AM, Chan KC. Calculated stress-shielding in the distal femur after total knee replacement corresponds to the reported location of bone loss. J Orthop Res. 1996; 14:778–85.
28. Kettelkamp DB, Jacobs AW. Tibiofemoral contact area: determination and implications. J Bone Joint Surg Am. 1972;54:349-356.
29. Walker PS, Erkman MJ. The role of the meniscus in force transmission across the knee. Clin Orthop Relat Res. 1975;109:184-192.
30. Sweigart MA, Zhu CF, Burt DM, DeHoll PD, Agrawal CM, Clanton TO, et al. Intraspecies and interspecies comparison of the compressive properties of the medial meniscus. Ann Biomed Eng. 2004; 32:1569–79.
31. Fithian DC, Kelly MA, Mow VC. Material properties and structure-function relationships in the menisci. Clin Orthop Relat Res. 1990; 252:19–31.
32. Fukubayashi T, Kurosawa H. The contact area and pressure distribution pattern of the knee. A study of normal and osteoarthrotic knee joints. Acta Orthop Scand. 1980; 51:871–9.
33. Ahmed AM, Burke DL. In-vitro measurement of static pressure distribution in synovial joints—Part I: Tibial surface of the knee. J Biomech Eng. 1983; 105:216–25.
34. Walker PS, Hajek JV. The load-bearing area in the knee joint. J Biomech. 1972; 5:581–9.
35. Thompson WO, Thaete FL, Fu FH, Dye SF. Tibial meniscal dynamics using three-dimensional reconstruction of magnetic resonance imaging. Am J Sports Med.1991;19:210-216.
36. Ricklin P, Ruttimann A, Del Bouno MS. Diagnosis, Differential Diagnosis and Therapy. 2nd ed. Stuttgart, Germany: Verlag Georg Thieme; 1983
37. Fairbank TJ. Knee joint changes after meniscectomy. J Bone Joint Surg Br.1948;30:664-670.
38. Voloshin AS, Wosk J. Shock absorption of meniscectomized and painful knees: a comparative in vivo study. J Biomed Eng.1983 ;5 :157-161.
39. Krause WR, Pope MH, Johnson RJ, Wilder DG. Mechanical changes in the knee after meniscectomy. J Bone Joint Surg Am. 1976;58:599-604.
40. Fukubayashi T, Kurosawa H. The contact area and pressure distribution pattern of the knee: a study of normal and osteoarthritic knee joints. Acta Orthop Scand.1980;51:871-879.
41. Kettelkamp DB, Jacobs AW. Tibiofemoral contact area: determination and implications. J Bone Joint Surg Am.1972;54:349-356.
42. Jones RE, Smith EC, Reisch JS. Effects of medial meniscectomy in patients older than forty years. J Bone Joint Surg Am.1978;60:783-786.
43. Kurosawa H, Fukubayashi T, Nakajima H. Load-bearing mode of the knee joint: physical behavior of the knee joint with or without menisci. Clin Orthop Relat Res.1980;149:283-290.
44. Seedhom BB, Hargreaves DJ. Transmission of the load in the knee joint with special reference to the role in the menisci: part II. Experimental results, discussion and conclusion. Eng Med.1979;8:220-228.
45. Radin EL, Rose RM. Role of subchondral bone in the initiation and progression of cartilage damage. Clin Orthop Relat Res.1986 ;213:34-40.
46. Arnoczky SP. Gross and vascular anatomy of the meniscus and its role in meniscal healing, regeneration and remodeling. In: Mow VC, Arnoczky SP, Jackson SW, eds. Knee Meniscus: Basic and Clinical Foundations New York, NY: Raven Press; 1992:1-14.
47. Markolf KL, Mensch JS, Amstutz HC. Stiffness and laxity of the knee: the contributions of the supporting structures. J Bone Joint Surg Am.1976;58:583-597.
48. Shoemaker SC, Markolf KL. The role of the meniscus in the anterior-posterior stability of the loaded anterior cruciate-deficient knee: effects of partial versus total excision. J Bone Joint Surg Am.1986 ; 68 (1):71-79.
49. Musahl V, Citak M, O’Loughlin PF, Choi D, Bedi A, Pearle AD. The effect of medial versus lateral meniscectomy on the stability of the anterior cruciate ligament-deficient knee. Am J Sports Med. 2010 ;38 (8) :1591-1597.
50. Arnoczky SP, Warren RF, Spivak JM. Meniscal repair using exogenous fibrin clot: an experimental study in dogs. J Bone Joint Surg Am.1988;70:1209-1217.
51. Bird MDT, Sweet MBE. Canals of the semilunar meniscus: brief report. J Bone Joint Surg Br.1988 ;70 : 839.
52. Bird MDT, Sweet MBE. A system of canals in semilunar menisci. Ann Rheum Dis.1987;46 : 670 – 673.
53. Karahan M, Kocaoglu B, Cabukoglu C, Akgun U, Nuran R. Effect of partial medial meniscectomy on the proprioceptive function of the knee. Arch Orthop Trauma Surg.2010;130:427-431.
54. Skinner HB, Barrack RL, Cook SD. Age-related decline in proprioception. Clin Orthop Relat Res.1984;184:208-211..
| How to Cite this article:. Chawla A, Aggarwal AP. Menisci: Structure and Function. Asian Journal of Arthroscopy Aug – Nov 2016;1(2):3-7 . |